|
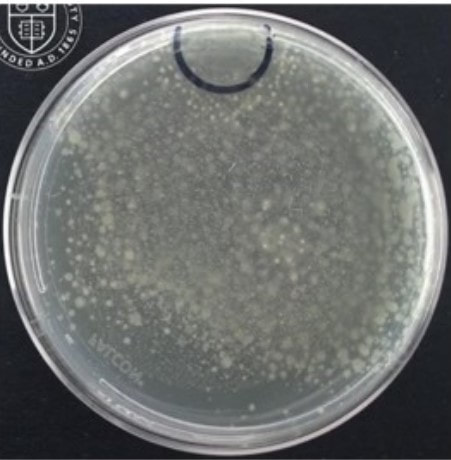
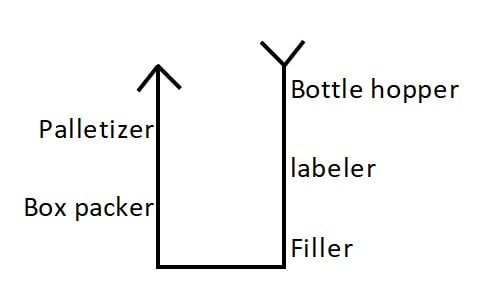
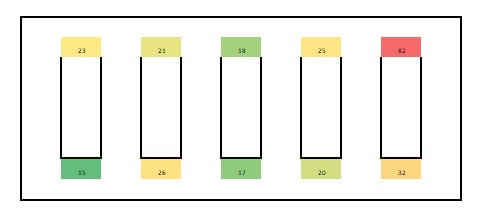
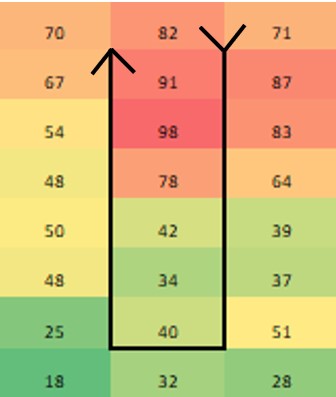
Paenibacillus glucanolyticus. I don’t know how every facility doesn’t have to deal with this bug. The bug is pictured below. Any microbiologist would call this plate TNTC (too many colonies to count / >250 CFU/plate). This organism drove me crazy when I was the Plant Hygienist at a soap manufacturing plant. I referenced this event in my previous post about my recent job interview presentation. This issue also fits my series of frustrating quality problems, so I want to expand on it. Our viable air monitoring recovered P. glucanolyticus every other month near a filling line. It was always TNTC! We were required to open a tedious investigation in our quality system each time. How do you find a specific microbe source in a non-controlled warehouse staffed by hundreds of minimally gowned personnel? Let’s set the stage. Imagine a typical warehouse. 40 ft ceilings, exposed steel rafters, vents, and piping. Stamped concrete floor with painted cement brick walls. Active forklifts and whirring equipment with semi-filtered exhausts blowing air in all directions. Hundreds of employees wearing uniform pants and long sleeve shirts. Hair nets and beard covers on everyone, but nobody wore gloves unless directly working with a filler. (Side Note about this environment) There were multiple filling lines in this warehouse. They were “U” shaped like the picture below. Bottles went from a hopper onto the filling line. They were labeled, filled, then packed in boxes. At the end of the line, the boxes were palletized. Environmental monitoring didn’t have specifications, but we set action levels based on past data. Higher results signaled something wasn’t normal. The picture below represents multiple lines in the warehouse. We monitored opposing sides of the lines (the colored spots). Most recoveries were "normal" growing bacteria. We had high Paenibacillus counts in the red area. What was strange- we only got TNTC results if P. glucanolyticus was on the plate. The counts were much lower if it wasn’t there. If the organism was prevalent enough to cause TNTC results, shouldn’t it show up at counts between 1 and 250 sometimes? I found interesting research about the organism. When growing on media, individual cells can migrate away from a starting colony and form their own colonies. When the plate is read 5 days later, the colonies will look completely distinct, like the picture at the top of this post. When multiple cells do this, and multiple cells from those colonies repeat the process, your single CFU at the onset looks like hundreds of colonies after incubation. I’ve since found a time-lapse video that shows this effect. A research team at Cornell created it. They placed a few CFUs in the small circle at the top of the plate. You can see how the organism grows and what the plate looks like at the end. Definitely not the traditional growth pattern with all generations contained in the same colony. I really wish this video existed when I struggled with this organism: Knowing this- knowing your TNTC result was probably representative of only a few CFUs- what do you do next? Can you just say the TNTC result isn’t real and stop the investigation? Unfortunately, we didn’t have time-lapse footage of our plates, so we couldn’t ignore the results. Any individual plate could have been caused by TNTC CFUs. We even had this organism show up as TNTC on a product plate, so we had to discard a batch due to it. This info about the organism made my investigation harder. I couldn’t just look for obvious signs of mass contamination in the area. I now had to find a source in this giant warehouse where the organism was merely surviving. Luckily, a week of expanded EM around the line pin-pointed the source. We collected air samples from multiple locations around the line. We sampled each of those spots every shift for 7 days, averaged the results (setting TNTC to 250), and made a heat map like this: The bottle unscrambler was in the hottest spot. This chaotic machine somehow transferred bottles from the hopper to the filling line by spinning them around really fast. A maintenance mechanic helped me take it apart and douse it in sporicide. I was told maintenance cleans the unscrambler during the yearly PM, but the presence of a type of bottle that hadn’t been used in 3 years in some of the crevices determined that was a lie.
I worked at this company for another year after cleaning the unscrambler. We didn’t see the organism again during that year, so I’m confident this cleaning (plus the PM cleaning instruction update) was effective. Of course, this organism popped up as TNTC again at the next company I worked for. How have I never heard other Micro labs struggling with it?
0 Comments
Microbiologists working in pharma cleanrooms may be horrified picturing this environment. Every product must be contaminated!
This set up is very normal for non-sterile consumer goods. It’s important to note - we did bioburden testing on three samples from every lot made in this warehouse. Our recovery rate - the percent of samples with any micro recovery at all – was less than 2%. That stat includes:
Microbes getting into the product from this open environment was extremely rare. (atleast, microbes that we were able to recover during testing). This knowledge heavily influenced my micro contamination investigations when I returned to pharma manufacturing. Investigators rushing to close their investigation record were quick to blame micro excursions on (heavily gowned) personnel in the area or imperfect cleaning around the time the sample was collected. My time at the soap warehouse taught me those factors aren’t as likely to contribute to an excursion as most microbiologists think. As an extra note- I was proud of the quality assurance at this soap factory. I was especially proud when we made the conservative decision to reject the lot associated with this P.glucanolyticus organism. It was an expensive decision to make. To show my pride, the soap we made was my christmas gift to my extended family the year I worked there. I gave the same gift in the wake of Covid in 2020. We were a contract manufacturer and didn’t own the products we made, so I had to pay full price for all of this. Let's get you back to the original post. This is the "20 Minute" presentation I gave during my last job interview. I was asked to prepare a presentation about the benefits of genotype identification to a laboratory lead that uses phenotype IDs. I converted the slides and my script/notes into this post. The presentation is pretty bland due to the limited time to make it. This post is LOOONG. and pretty boring. But I think the examples I give in slides 13-16 are pretty interesting. Slide 1: Good Morning! My Name is Jonathan Kallay. Today I’d like to talk with you about your current Microbial identification process and some opportunities for improvement. Slide 2: I’ve spent the last 12 years in Micro laboratories. I’ve worked with a variety of regulated products. From consumer goods and lab supplies to biopharmaceuticals and small molecule parenterals. I specialize in non-conformance investigations. Slide 3: From my understanding, you primarily use phenotype identification methods. These methods are often more affordable than Genotype identification in the short-term. However, an investment in genotype testing can improve laboratory and manufacturing performance, leading to significant cost savings in the long run. Slide 4: Here’s a quick run-down of what I want to talk about today:
Slide 5: Phenotype identification is most commonly performed with a combination of colony morphology analysis, Gram stain, and biochemical enzyme reactions. An analyst will inspect a colony on a test plate to determine if the organism is most likely a mold or bacteria. If the organism is a bacteria, it will be subcultured with a gram stain performed on the subculture the following day. Depending on the gram stain result, different biochemical enzymatic tests can be performed to obtain an organism identification. Slide 6: These steps can be pretty effective to get a general understanding of the organisms in your environment.
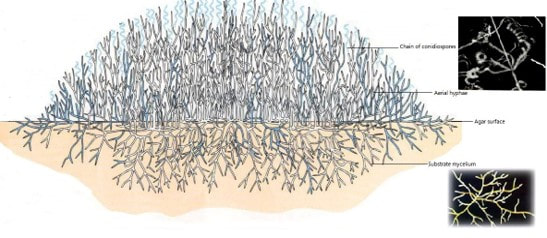
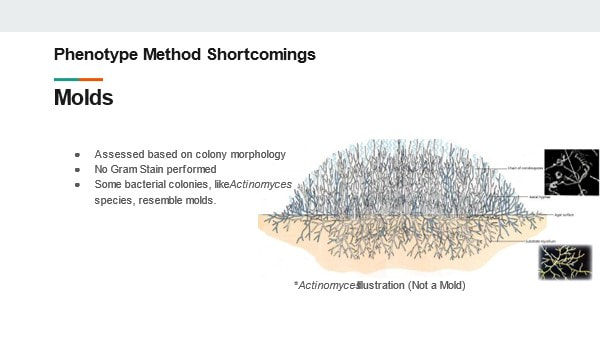
These procedures have been in use for decades. They’re generally considered accurate and have historically been acceptable in the eyes of regulatory bodies. But we need to understand why these tests are insufficient in today’s manufacturing environment Slide 7: The first short coming I want to talk about: Molds! Why are molds a problem? We determine they’re mold based on how the colony looks. No gram stain is needed. We can even perform some genus level identifications by inspecting the hyphae and fruiting bodies. But not gram-staining IS the problem- There are bacterial colonies that look like mold. For example- Actinomyces species. They grow branches of hyphae just like a fungus. And the name- “Myces” comes from the greek for fungus. It has fooled people looking at it for hundreds of years. If this relatively common organism becomes prevalent in your environment, your search for the source will have you looking for mold, instead of the anaerobic environments these species grow best in. Slide 8: Gram Stains In my experience, the gram stain is the most common phenotype test used in the microbiology lab today. The gram stain result is used for trending and identifying the general source of the organism. It’s also a prerequisite for running biochemical tests. But right off the bat there is a laundry list of things that can go wrong during the gram-stain:
If any of these issues occurred, they’d almost exclusively be blamed on analyst error. Regardless, once the result is generated it’s almost impossible to determine if an error occurred. When a result is generated by an analyst’s interpretation, there is little opportunity to review and verify the result. In the Lab, Data Integrity is the ability to document and verify objective test results. We don’t have this ability for most gram stain methods in use today. Slide 9 But I want to give the benefit of the doubt that testing issues have been worked out with iron-clad procedures and meticulous technique. Analysts are perfectly trained and able to perform all gram stain steps and readings correctly. That still leaves us with some inherent problems with the test: Gram variable bacteria! They vary in the stains they absorb and retain during the test depending on where each cell is in its growth cycle. That can be the difference between ID’ing the organism as gram positive or gram negative. Even with perfect gram-staining technique, the test can yield both purple and pink cells on your slide. This is common for many Bacillus, Clostridium, and even those same Actinomyces species. Depending on how those colonies are growing on gram-stain day, you could generate either a positive or a negative result for the same species. There are also organisms that vary in shape throughout their life cycle. A common genus is named for this phenomenon- Coccobacillus. These short-rod species appear as little balls (cocci) under a microscope. This is bad news when the laboratory makes an identification decision based on this appearance. The organism will be ID’d as a cocci when the organism shares most other characteristics (like where you're mostly to find it) with other rod species. Let’s not forget, there are some bacteria where gram stains just don’t work. Notably Mycobacterium species. The stains can’t get past the cell wall. Missing the identification for these organisms would be devastating for certain biologic pharmaceuticals. Slide 10: Biochemical tests also fall under the umbrella of phenotype identification. These tests can improve the accuracy of our target organism identification. However, these have some similar technical laboratory issues common for gram stains. For example, something as simple as the catalse test leaves room for interpretation. After mixing hydrogen peroxide with a cell culture, bubbles may appear. Bubbles signal the organism is catalase positive. But what if you see only a few bubbles? Or what if the bubbles originated from physically mixing the peroxide with the culture? This is another subjective measurement that laboratories should strive to eliminate. The way I see it, the bigger problem with these tests is they’re often only as good as your gram stain result. You may determine your organism is gram-negative rod, oxidase positive, and catalase negative (among other enzymatic test results). If the organism was actually a gram-positive rod due to one of the issues we previously discussed, then you assigned an incorrect ID. Even some prominent automated test systems that perform biochemical testing rely on your gram stain result. The gram stain result determines the cartridge you place your organism into for those systems. Slide 11: To re-hash the shortcomings of these phenotype tests:
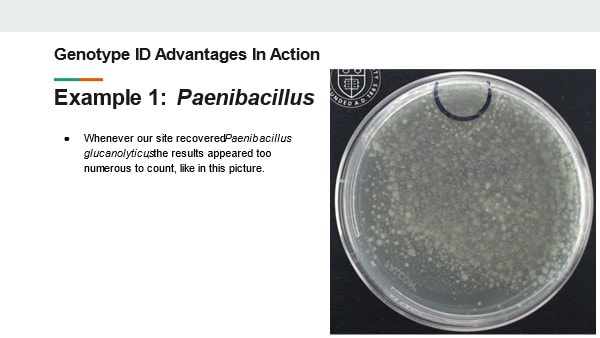
Slide 12: How does genotype testing account for these shortcomings? The answer depends a little bit on the test method used. There are methods that sequence Neuclic acids from the organism. There are also methods that interact with protein in the cells to reflect a protein fingerprint. Both methods compare the results to a library of known, validated organisms. All test results are determined based on objective measurements correlating to the specific nucleotide sequences or protein concentrations in the organisms. The test work for any microorganism. This eliminates any concerns that a subjective test could impact the result. Better yet, the tests are sensitive enough to identify to species and subspecies levels. How can such precise identifications help you? Well, I’d like share a few times how it helped me in my career. Slide 13: Example 1 In 2015 my laboratory was plagued by a Paenibacillus species. A few environmental monitoring and product test plates looked like this. The result was too numerous to count. We identified this organism to a species level and learned some really interesting things about how it grows. Slide 14: This video illustrates our findings. You should be able to see a small black circle on the top of the plate. An inoculum (a few CFUs) of Paenibacillus glucanolyticus was placed in that circle. As the video plays, you can see colonies pop up all over the plate. By identifying this organism down to the species level, we learned the most likely reason for the TNTC results wasn’t because we had mass contamination in our sample. We just had an organism that grows ... weird. This growth made 1 CFU appear as hundreds. This revelation was critical for us as we determined our investigation and response strategy. The TNTC results weren’t due to massive contamination of our product or environment. We were able to target our search for the organism in more nuanced locations. Best of all, we didn’t need to discard product or shut down the manufacturing line. Slide 15 Example 2: Another laboratory event from my past caused multiple tests to be contaminated with Bacillus Subtilis. This is one of the most common species that our lab recovered from around the facility. Locating the contamination’s source was going to be a nightmare! We collected multiple investigational swabs and found B. subtilis on lots of them. However, only one matched the strain identification from our original test samples - It came from the glove we used to remove items from the autoclave. Had we not had the strain identification, this is what would have happened:
With the genotype strain identification, we found the exact source of our problem. We changed our glove to one that was easier to clean. We never had the issue again. This saved us so much time and resources by minimizing repeat investigations and cleaning. Slide 16: Example 3: A few years ago I worked at a biopharma manufacturer. The manufacturing rooms were kept cold (around 2-8C) to prevent protein de-naturing. This conveniently made it harder for microbes to reproduce in these rooms. That didn’t stop a Pseudomonas fluroescens species from contaminating our in-process product. Due to the organism being GNR and the amount of water used in the process, we believed the organism was coming from the water system. We were running extra, expensive, time consuming hot water sanitization cycles to flush this organism out. However, with a full genotype strain identification, we found the organism was a subspecies in the Pseudomonas fluorescens group called Pseudomonas antarctica. If you can’t tell from the name, this organism thrives in cold environments. This identification helped us re-focus our efforts on areas in the cold room where the organism could colonize. We ultimately matched the organism to the exact subspecies in a set of manufacturing hoses. Slide 17: Those are only a few examples from my career where a good identification program changed the course of our investigation. As you can see, the investment saved us lots of time AND money. The genotype IDS saved us from:
Luckily, all of these sites were using (or transitioning to) genotype identifications during these events. They clearly reaped the benefits from this process. Slide 18: But the original decision to implement the identification program was based on the trends of regulatory expectations for environmental monitoring programs. There are hints in regulatory guidance documents, warning letters, and other industry writings on the topic that agencies are expecting more from environmental monitoring programs than they have in the past. The EMA’s Annex 1 Expects all organisms recovered in grade A and B clean rooms to be identified to a species level. The FDA agrees in A 2004 guidance for sterile manufacturing . The FDA writing suggests the identification should be sensitive enough to match organisms if recovered in final product samples. The document goes so far as to call out the superiority of Genotypic methods over phenotype 8techniques: “Genotypic methods have been shown to be more accurate and precise than traditional biochemical and phenotypic techniques. These methods are especially valuable for investigations into failures (e.g., sterility test; media fill contamination). Slide 19: A few final notes from regulatory documents There are non-binding instructions to ID mycoplasmas to a species level In a 2010 FDA guidance for the Vaccine industry. There are similar strain typing suggestions to ensure contaminants aren’t originating from any biological sources. And these regulations aren’t just limited to the sterile industry. In food manufacturing, the FDA currently uses genotypic testing during site inspections when there are suspicions of food borne illness. Throughout all these industries data integrity concerns (similar to those with phenotype ID test methods) are becoming a more frequent target for FDA warning letters. As you can see, Having the capabilities to proactively perform genotype testing can save time and help with a decisions when facing a recall or other regulatory action. Having the capabilities to proactively perform genotype testing can save time and help with a decisions when facing a recall or other regulatory action. Slide 20: The ultimate decision to invest in genotype testing can be difficult. There’s a comfort level in sticking with what you believe is already working. Hopefully you can fully appreciate all the ways that genotype testing can work better for you, your laboratory, and your customers. Slide 21
|