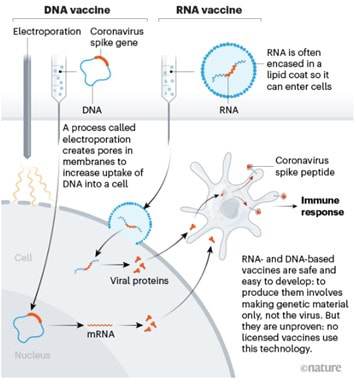
|
I got flashbacks reading about the technology used for coronavirus vaccines. What’s my connection to the vaccines? The lipid membrane delivery system. It’s a really cool bio-technology used for drugs that need to get into certain cells to work, but the drug has issues getting into those cells. The lipid membranes are made of the same molecules as our cell membranes. The cell absorbs the lipids and lets the drug inside just like the picture below. Follow the RNA vaccine path from the top right. There’s a more intense description in the article I stole the picture from: I worked with a chemotherapy drug that used a membrane like this. The generic version of the drug, which consisted of just the active ingredient with no membrane, had intense side effects. The side effects were drastically reduced with the lipid membrane delivery system. Instead of the drug running rampant in the blood stream, it was delivered directly into the cells of the target organ. It’s an amazing medicine.
When I started working in a micro lab, I was trained to test this drug for bioburden. If you don’t know what bioburden testing is or why we do it, this should catch you up. For a significant number of lots, the liquid drug product wouldn’t filter and the test wouldn’t work. The goal of filtering is to retain all cells on the filter. The problem with the lipid membranes- they form mini cell-like structures, almost on the same scale as the microbes we’re trying to retain. We’d run the vacuum powering the filtration process for hours at a time. A small amount of liquid slowly passed through the filter. The rest sat in the filter funnel indefinitely. The problem was hidden for years. Eventually, analysts determined the test could not be performed for some lots. At first, small investigations were written for these lots, treating them as one-offs with minimal risk. Ultimately, the analysts made it known they were having a problem and prompted a serious investigation. You may have a lot of questions on how we let this issue fester for so long. The general answer: this was the type of company that was responding to an FDA warning letter when I started and received a consent decree 5 years later. But here’s some general notes about the whole problem:
In the end, we never got closure on what exactly caused the filtration issues. Regulatory agencies required 100mL samples of this product tested for 2 different media types. We updated the method to allow for adding the results from multiple test plates based on how much product would pass through each filter. Sometimes only 5-10mL would filter at a time. Other times all 100mL filtered smoothly. The wording of the test method had to be flexible, but it was hard to interpret, just like how I’m struggling to describe it succinctly now. I felt like a failure after this investigation. I got no closure. I also don't have a good way to wrap up this little story, so we can both feel frusted. Now, I hope these lipid membranes aren’t causing similar issues for vaccine bioburden testers around the world.
1 Comment
Bioburden testing is performed on In-Process pharmaceutical products (or any non-sterile product) to quantify and identify micro contamination. The test is performed on a product sample prior to filtration, filling, and any final sterilization cycles. The test lets manufacturers know what types of organisms, and how many, are getting into the product before they’re physically removed or killed off. If they’re physically moved or killed off, why do we need to know if they’re in the product at that stage? Great question!
Bioburden testing is pretty straight forward:
There are other methods, but this was used for the product I talk about in the unfiltered story. It was the preferred method in the lab at the time. We could test large volumes, as opposed to the pour plate where we could only do 1mL a plate.
But how do we know microbes in the product will show up during this test? The drug may prevent the organism from growing or could even kill the microbes we’re looking for. That may not seem like a big deal – “the drug kills organisms, so we don’t have to worry so much about removing them!”. But re-read the bullet points and you’ll remember why we need to know if organisms were ever there. Back to the question- we know the test works because we validate it. We inoculate a sample with target organisms, test it, then confirm recovering those organisms after incubation. If we do this over multiple lots and recover about as many organisms as we inoculated, we consider the method validated. Other testing kinks get worked out during the validation too. What type of rinse too use, how much rinse, the material in the filter, etc… Hopefully this background helps you understand the test so you can relate to the problem we had. Let’s get you back to the investigation story! |