|
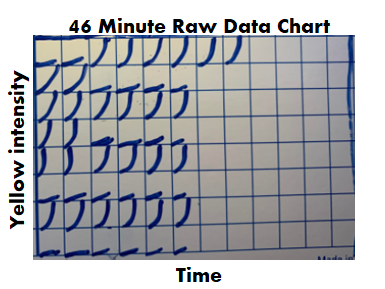
Part 1 of this series discussed endotoxin contamination in a WFI system. During the investigation, we noticed something strange in our historical data review. A couple city water samples didn’t have their expected endotoxin. Part 2 of this series looked into raw data for those tests. For one of those two tests, endotoxin levels were just below the limit of detection threshold. That made sense. I ended part 2 saying “I saw _______ ”. I wasn’t leaving you with a blank to fill in. I literally saw a flat line. What’s so interesting about a flat line? Quick recap- The lysate we used for testing (i.e. purified horseshoe crab blood), turns yellow over time. It turns yellow faster when combined with endotoxin. The test measures how fast samples turn yellow by collecting yellow intensity readings every couple minutes. After most tests, the raw data graphs look like this: Part 2 really goes into detail on this, but the squares are graphs representing each well of a 96 well plate. The right half of the plate didn’t have anything in the wells, so the raw data graphs look like empty boxes. They're actually flat lines running along the x-axis. The raw data for the 2nd atypical city water day looked like this: Tests wells with lysate, but no endotoxin, should have graphs like this: For the test in question, the graphs looked like lysate was never added to them, like this: Part 3 of this endotoxin series starts here. We found a single assay that looked like lysate wasn’t added to test and negative control wells. If that was truly the case, the samples were never tested, but the assay was guaranteed to have all results meet specification. The official test report can't detect this error.
So what do we do next? A bunch of things simultaneously. Identify our scope. It would’ve been easy to write this off completely. The raw data wasn’t used for any official purpose. We even reached out to the equipment supplier and they couldn’t support any conclusions from the raw data. According to them, if all parameters in the final report met requirements, the assay was acceptable. It also would’ve been easy to call this a one-off assay with (potentially) invalid results. We could have written up a low-impact non-conformance report and called it a day. But I’m proud my company supported the search for, and reporting of, everything impacted by this issue. Further review determined a single analyst was responsible for 3 months of assays with this pattern. We immediately considered them all invalid. Lysate was not used in the test wells of:
Damage Control The clock was ticking. Per 21 CFR 314.81(b)(1), the site was required to submit a Field Alert Report (FAR) to the FDA within 3 days. We had information concerning significant quality problems with distributed drug product. We had dozens of lots on the market without a valid, required relase test. To make matters worse, this site received a warning letter for endotoxin issues less than a decade prior to this event. It was all hands on deck in the endotoxin lab. We had 3 days to generate valid tests for all impacted lots. We tested retains for all of them. We barely had enough test equipment to complete all samples that quickly, but we somehow got it done. A non-conformance event was opened to address the impact of the water sampling that couldn’t be replicated. We also had to re-do equipment validations. We got through it, and I feel as confident as I can be in the quality decisions that were made during this time. Handle a Human Resources Nightmare. Officially, the analyst consistently repeated an error in their technique. This error somehow developed after months of testing correctly. The analyst added lysate to standard and positive controls wells first. These wells are supposed to have endotoxin - they require a lysate reaction for the final report. The analyst claimed adding lysate to these wells first helped prevent contamination (that in itself is faulty logic). Then, the analyst “forgot” to add lysate to the test wells. There were other raw data inconsistencies that contradict the official story. A variety of different colored products were tested over these three months. These products all have different “baselines” - the first reading when the equipment starts measuring yellow intensity of the sample. However, in assays where the analyst claimed to test these products, the baseline readings for the sample positive controls (where we know lysate was added) were the same as the baselines for water positive controls. The baseline should have been much higher for a colored product. To me- that meant the analyst never even attempted to test the product. I dug into all assays this analyst performed. I also checked on multiple assays performed by other testers to confirm this wasn’t a department wide issue. I came to the following conclusion: The analyst knowingly tried to cheat the test to prevent “complications”
I wasn’t involved in the HR and management discussions. I believe they agreed with my conclusions, but they officially aligned with the analysts’ explanation of events. I think they were stuck:
Prevent this from ever happening again. We made raw data review an official process moving forward. Every analyst was trained on what to look for when performing and reviewing assays. There were strange nuances though. For example, if a bubble formed during pipetting, it could impact the baseline reading and cause a flat line graph. There were ways to tell if that was the case, but impossible to define in an SOP. Clearly the analyst never should have made this error. I think the stress of the job contributed to it. The job occurred on 2nd shift. This shift was understaffed and often dealt with time sensitive samples. The samples could come at any time during the shift. A minor issue with an assay at the end of the shift could require the analyst to work unpaid (salaried) overtime through the night. There was no 3rd shift in the lab to help on those late nights. The temptation to cut corners proved too much. Soon after this event, a full-time 3rd shift position was created. It was extremely comforting knowing critical samples could be handled as a team. It removed a lot of the corner-cutting temptation and significantly reduced the stress on the job in general. This was such a strange event it never would've repeated regardless of the actions we took. But the Lab became a better place because of those actions.
0 Comments
Leave a Reply. |