|
Welcome back to the final segment of this series! You can catch up on Part 1, Part 2, Part 3, and Part 4 <-- right there! To sum up all 4 of those parts- We solved a Bioburden problem! Product contact piping wasn’t getting cleaned between runs. It seems so easy in hindsight. All we had to do was adjust the cleaning cycle. How could we mess that up? That’s exactly what I’ll go into in this final part of the series. We went a year before finding our biggest mistake. I was instructed to close the investigation when we figured out the bacteria’s source. Then, engineering opened a second investigation to document why the piping wasn’t getting cleaned. Their corrective action plan wasn’t too complicated. They updated cleaning cycles to:
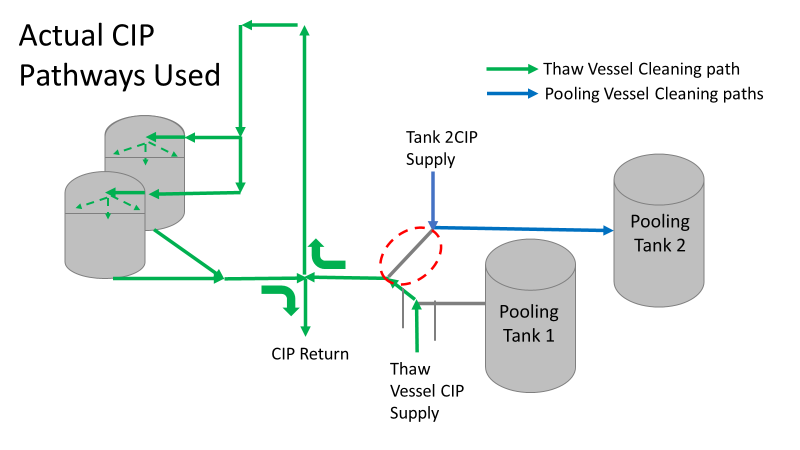
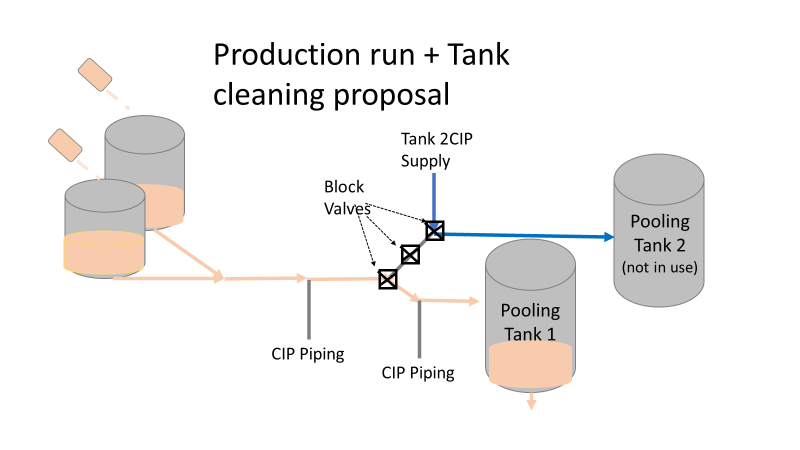
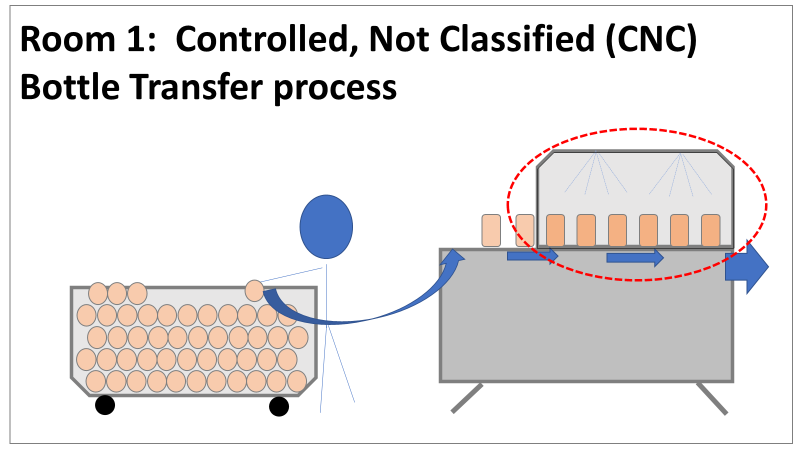
So what happened a year later? We got another bioburden OOL at the same manufacturing step. This time it was a hodge-podge of gram-negative water organisms. Mostly Stenotrophomonas. The old gang got back together to look it over. Site management followed this one even closer, knowing we were now delaying active commercial runs. The Engineering manager confirmed CIPs were performed before the run. The team had another equipment inspection and swabbing party. We found lots of gaskets in poor condition, so we replaced them and scheduled a contract cleaner again. This was major déjà vu, so I reached out to the engineering techs. I wanted to learn more about the cleaning programming. Our talk was all we needed to find the new root cause. For the entire year, the site was only using one of the pooling vessels. A few runs before this OOL result, the qualification for the second pooling vessel was complete, so it started to get used. Although the operators could now choose the appropriate CIP inlet, there was no procedure to prompt operators on which one to use. The CIP cycle continued to run through the same inlet. When the 2nd pooling vessel was used, the same section of piping wasn’t getting cleaned (circled in red below). This was also fixable! The operating system required a cleaning cycle to be performed before a tank could be used. We designated that circled section of piping to register like a tank. The system would electronically “block” that section from being used if it wasn’t cleaned. We would never have this problem again! This problem would have easily been identified had we walked through the process from an operator’s perspective the first time we “solved” the bioburden issue. But there were other system problems that should have been solved with the first investigation. Quality improvements are normally seen as extra costs and delays. But these quality improvements can go hand in hand with manufacturing efficiencies. I have two examples from this critical investigation. Manufacturing efficiency example 1: Operators were frustrated with cleaning delays. They couldn’t clean tank 1 while the system was running to tank 2 (and vice versa). This didn’t make sense. The equipment was designed to allow for that. There were multiple block valves between the CIP inlets and the opposing product lines. This problem became my green belt project. I left the company before the change was completed, but we had a plan in place when I left. In the scenario above, the production run “owned” those 3 valves. This prevented any other cycle that could use those valves from starting. All we needed to do was update ownership of the valves in those cycles, and we could save the production team 3 HOURS between each run. This was huge for a site planning to increase from 10 to 14 runs per week. With the experienced group of engineers working on the bioburden issue, this delay could have easily been fixed during the initial investigation. This was another example of not looking at the system through the lens of the people using it. Manufacturing Efficiency Example 2. I discussed in the earlier parts of this investigation that the microbiologists were pre-occupied with the bottle transfer process. The bottle rinsing chamber caught their eyes. The rinse chambers had open-able windows lined with gaskets. During the investigation, the gaskets were removed to swab their housing. The swabs were covered in black residue. There were no bacteria in any of these samples. However, the residue was so concerning that we made a major update to the chamber cleaning process. The new SOP required gaskets to be removed, cleaned, and placed back into position between each run. This added a huge extra delay to the cleaning process. There were 12 gaskets. It took a half hour to clean them all. The extra quality problem the cleaning caused was even worse.
The gaskets’ integrity deteriorated between each manipulation. Over time, they stopped forming a good seal. There were even times where operators replaced them backwards (this was very easy to do). This led to major leaks during runs, flooding the bottle transfer rooms. The initial investigation team added a time-consuming process that wore down equipment and caused leaks in the manufacturing area. The team was grasping at straws for a root cause. They pushed the change to feel like they were doing something to make the system cleaner each run. If you read (red/reed) part 3 of this series, you’ll see why they never should have considered that area as a source. Cleaning the gasket housing as part of the routine equipment PM would have been sufficient to address the residue issue. I created the SOP redline for the process owners to remove the gasket cleaning process. It was never given priority (likely because the process was stopped without changing the procedure). The site probably still has the cleaning instructions in their procedure. There are other compliance issues if an effective SOP is not being followed. The major take away from the last part of this series- walk through your processes from the point of view of your operators. This is text-book poka yoke. Poka yoke is often taught in a quality/error-proofing context. But when you’re given this opportunity to look for production improvements, use it! P.S.- Blaming gaskets for a micro issue is lazy. You can look at any piping system (with or without bioburden issues) and find gaskets in poor condition. Finding them doesn’t mean they’re the cause of the issue.
0 Comments
Leave a Reply. |